Periodicity
|
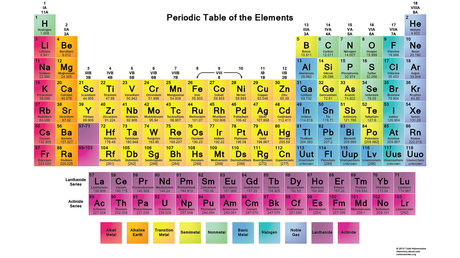
Periodic Table
Periodic Table Instructions Periodic Trends Powerpoint Periodic Trends Graphing Trends Activity Graphing Trends in Excel Instructions Periodic Table History Video Periodic Trends video Periodic Trends overview (Great Quiz Review) Periodic Family Labels- Alkali Metals-Group 1A Alkaline Earth Metals- Group 2A Lanthanides- Period 6 F-Block Actinides- Period 7 F-block Transition Metals- D-Block or all B Groups P-Block Metals- Al,Ga,In,Sn,Tl,Pb,Bi,Po Mettaloids- B, Si, Ge, As, Sb, Te Non-Metals- H,C,N,O,P,S Halogens- Group 7A Noble Gases- Group 8A Ionization EnergyIonization Energy-
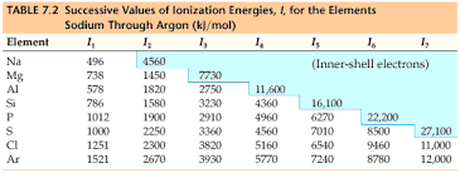
The energy required to remove an electron. The first ionization is the energy required to remove 1 electron, 2nd ionization energy is the energy required for the second electron, third etc. Understanding Ionization Energy-
You may determine the number of valence electrons and therefore the element by observing major changes in ionization energy. For example the ionization energy of Sodium in the above table increases from 496 to 4560. This is because taking the 1 valence electron requires much less energy than taking an electron from the full energy shell when it has a configuration like Neon. |
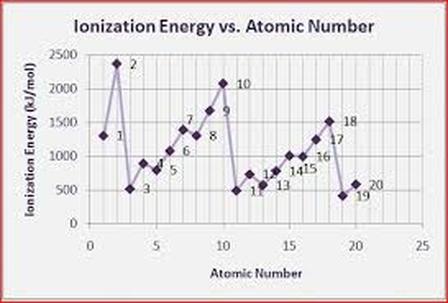
Periodic Trends Graphs:
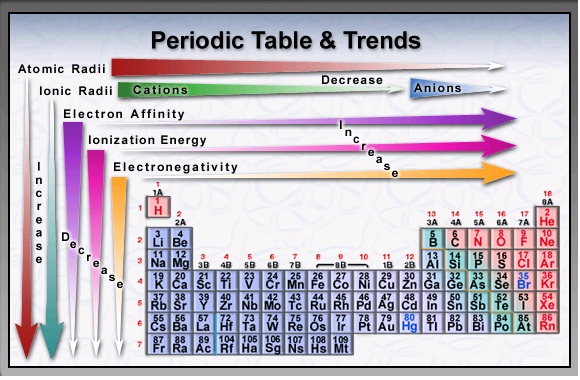
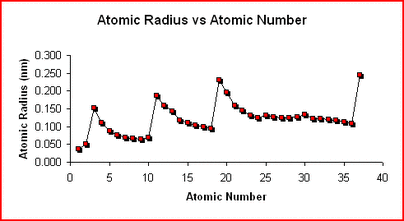
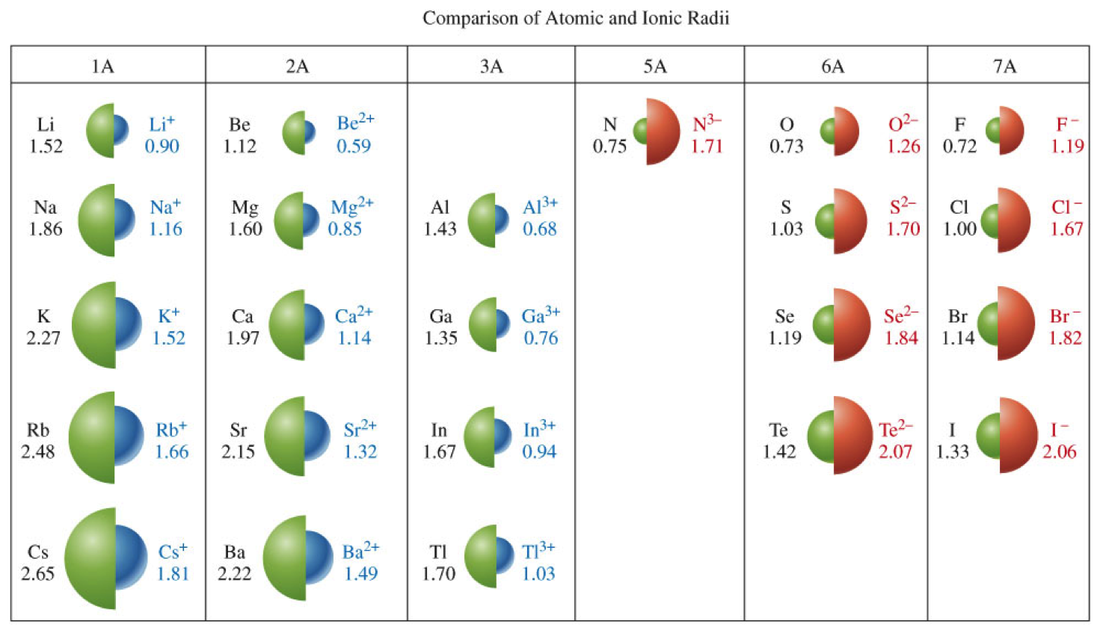
Atomic Radius and Ionic RadiusAtomic Radius-
This is essentially the size of an element. Francium has the largest atomic radius and flourine has the smallest atomic radius. The trend is the same for ionic radius as well, However negative ions become quite larger while positive ions become quite smaller as seen below. ElectronegativityElectronegativity-
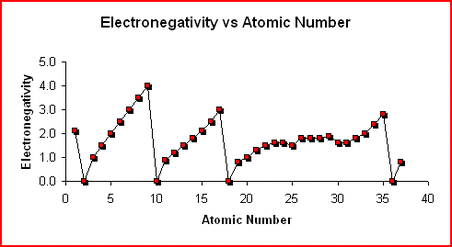
The likelihood of an element taking an electron from another element to form a negative ion. (Noble Gases have no value for electronegativity) Review Key |