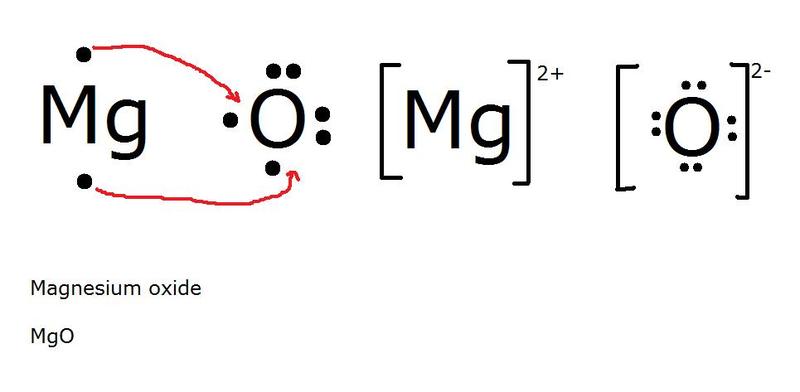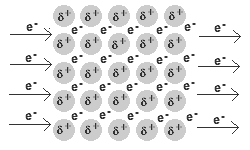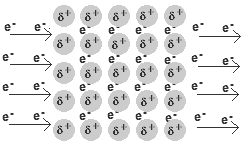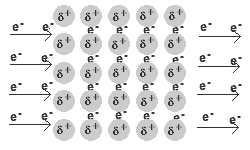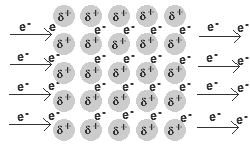Chemical Bonding
Unit 4: Ionic & Metallic BondingIonic-
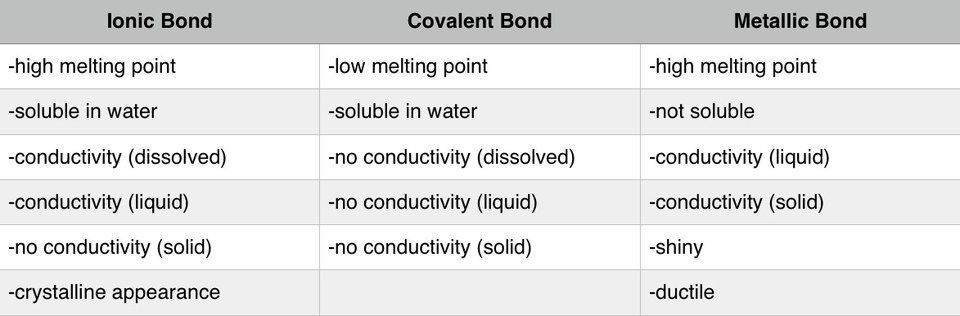
Ionic Bonds are Chemical Bonds between Metal positively charged cations and Nonmetal negatively charged anions. In Ionic Bonding Electrons are exchanged from the Metals to the Nonmetals. After the exchange of electrons Metals take on a positive charge making them a cation, and Nonmetals take on a negative charge making them an anion. This is due to the overall change in electron to proton ratio. These charges are held together then by electromagnetic force which makes ionic bonds the strongest chemical bond. Because of the nature of these bonds we observe specific chemical properties of ionic bonds. They tend to have a high heat capacity, meaning they are able to withstand pressure to break bonds. The breaking of bonds is often seen in changes of matter including boiling and melting points. For ionic bonds they have a high melting and boiling point because it takes a significant amount of energy to break these bonds. These are also soluable in polar solutions such as water. When in solution these ions also conduct electricity because of their charges. In nature you usually find ionic bonds in the form of crystalline solids often in rocks and minerals which are non-living parts of nature Metallic- Metallic Bonds are Chemicals Bonds between Metals and Metals. These Metals bond because of delocalized electrons from their valence shells as they attempt to become positive cations. These delocalized electrons constantly are attempting to bond with the cations as a "sea" of electrons because of electromagnetic force. However, the desire for cations to remain positive and remove electrons creates an electrostatic force between the electrons and positive Metal cations forming a bond.. Because of the nature of delocalized electrons the specific properties of Metallic bonds exist. It is solely based on the behavior of these special forms of electrons that these properties exist. Metallic compounds are the weakest bonds, but because they are constantly reforming they have a high heat capacity and boiling point. The free electrons also allow for electricity and heat energy to be conducted very well through metallic bonds. Because of the free nature of these electrons metallic compounds are also able to change shape while remaining a hard solid. These properties are known as malleability and ductility. Malleable meaning they can be bent and molded in specific shapes, and ductile meaning they can be stretched out into a wire.The arrangement of the cations into a crystalline form not only means metals are usually always found solid in nature but gives them a luster, or shine, that is unique to metals. (electricity conducting through a metal where delocalized electrons are displaced by metallic cations as extra electrons are introduced from electricity)
|
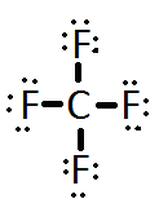
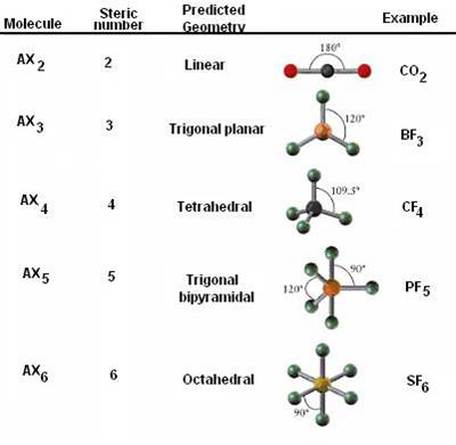
Unit 5: Covalent BondingCovalent- Covalent Bonds are Chemicals Bonds between Nonmetals and Nonmetals. These nonmetals both tend to become cations as ions so they both desire to take electrons to complete noble gas configuration which is usually 8 electrons but can be 2 in the context of Helium. Because both electrons desire to have more they share electrons to complete the needed total of electrons set by the octet rule. This sharing of electrons forms the bond between the two elements. The needed number of elements per compound can be found by completing the octet rule for each. Because these bonds are created by the sharing of electrons specific properties arise in covalent compounds. These are weak bonds because the position of the electron determines the strength of the bond at given moments. Covalent bonds occur naturally often as gases, organic compounds (carbon compounds), and nutrients necessary for living things. They are low conductors of electricity making them insulators this is because most nonmetals want to take any free electrons they can to form noble gas configuration. These compounds are also usually soft and brittle. Due to the weakness of this bond they also have low heat capacity causing a low boiling and melting point as the compounds change their state of matter. Resonance Structures- are structures of the same molecule that can have different arrangements of the molecule itself. NO2 for example has two different structures one where a double bond with oxygen is on the left side of the molecule and another where it is flipped and the double bond is on the right side of the molecule. VSPERCovalent Compounds VSPERhttps://www.youtube.com/watch?v=xNYiB_2u8J4-
Quizlet Template for Geometry- (E stands for valence pairs which cause polarity and The MG writing is the polar geometry when those extra electrons are greater or less than the other elements in the compound) AP CHEM Quizlet good Information on intermolecular forces and polar geometry YES some of this is TMI (too much info) Molecular ForcesIntermolecular Forces- Forces between molecules, Examples include Hydrogen Bonding> Dipole-Dipole> Van Der Waals Forces> London Dispersion Forces
They are listed in order of strongest to weakest above. Intramolecular Forces- Forces within "A" molecule such as the sigma and pi bonds and it's polarity. Polarity is determined by an unequal distribution of lone electron pairs. |