Unit 2:Atomic Theory and Structure
|
Objective Homework
Objective homework key -Objective I- Atomic Theory Scientist- Democritus- John Dalton- J.J. Thomson- Goldstein- Ernest Rutherford- Robert Millikan- James Chadwick- Niels Bohr- Werner Hiesenburg- Subatomic Particles- -Objective II- Protons- These are positively charged particles discovered by Rutherford/Goldstein based on the hypothesis of an equal positive charge to go with the negatively charged electrons previously discovered by Thomson. The proton is the basis for the Atoms atomic number and identity in general based on the properties that it carries. In a neutral atom the number of protons and electrons are the same. A hydrogen atom is essentially just a proton. Protons were found to be in the nucleus of atoms based on Rutherford's experiment. Neutrons- These are particles of no charge that were discovered by James Chadwick based on the question of the "mystery mass" of the atom after the discovery of the proton. Neutrons are a large player in nuclear reactions and chemistry and can be found in isotopes. Isotopes are singular examples of an element where they have a different number of neutrons usually making the element have a greater mass. The proton and neutron totals make up the atomic mass of the element. It is located in the Nucleus of an atom which was discovered by Ernest Rutherford and his Gold Foil Experiment. Electrons- These are particles of a negative charge and were discovered by J.J. Thomson through his Cathode Ray experiment finding that the particles in the Cathode ray were negative in nature and allowing for the minuscule mass of the electron to be found based on the ratio of charge to mass. The electron plays a large role in chemical bonding and reactions. The location of electrons is outside of the nucleus and based on a probability location due to charge and speed developed by Erwin Schrodinger. In the past it has been theorized to be in orbits, energy levels, and now orbitals(of probability). -Objective III- Isotopes Isotopic Notation An isotope is simply a particular atom that happens to have a different mass based on the number or neutrons in the atom. Practice Sheet -Objective IV- Electon Configuration- Electron configuration practice (quizlet) Orbital notation video Electron configuration notation video Noble gas configuration video Orbitals (s,p,d,f) (image) Key terms- Hund's rule, Aufbau Principle, Pauli Exclusion Principle, orbital, orbital notation, electron configuration notation, noble gas notation, energy level, Quantum wave equations- E=hv c=vλ Key terms- Planks constant, energy emission, electromagnetic spectrum, wave particle dilemma, wavelength, frequency(, speed of light constant (c), photon, Objective 4 Worksheet Objective 4 Worksheet key |
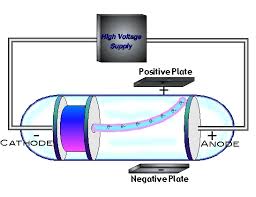
Cathode Ray Experiment ( JJ Thomson)
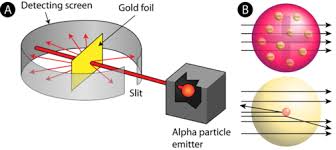
Gold Foil Experiement (Ernest Rutherford)
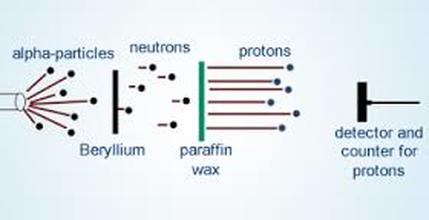
Berryllium Bombardment Experiment (James Chadwick)
Dalton's Model
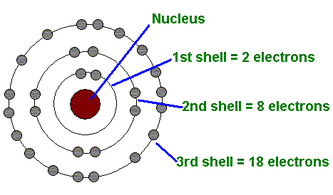
Bohr Model (Niels Bohr)
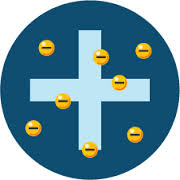
The Plum Pudding Model (Thomson)
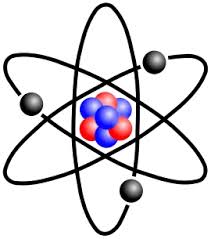
Planetary Model (Rutherford)
|